AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |
Back to Blog
Openoffice calc3/30/2023  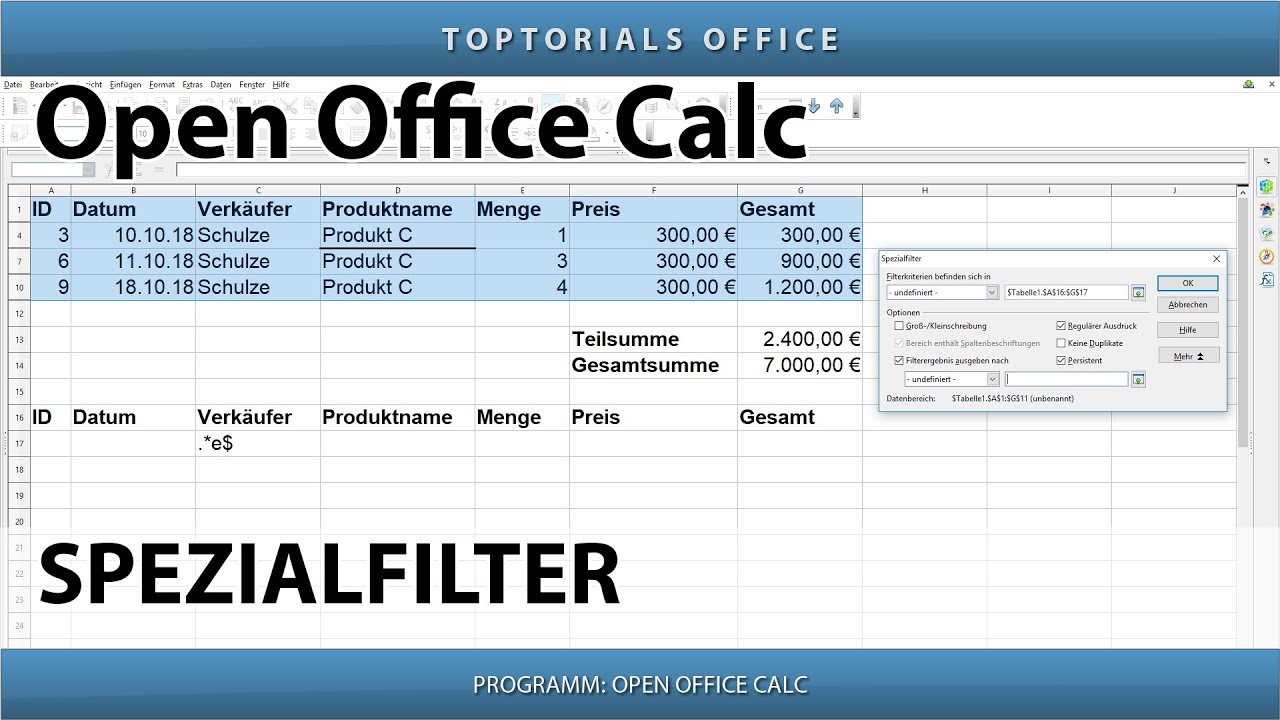
The Company focuses on discovering, developing, manufacturing, and commercializing new therapeutic cannabinoids in a wide range of disease areas.ĭanCann Pharma is EU-GMP-approved by the Danish Medicines Agency under the Danish Pilot Programme for medical cannabis, and have, among other things, license for manufacturing, export, import and distribution. DanCann Pharma is a licensed production and distribution Company. CannGros currently has three products (dried flowers and granules) included on the Danish Medicines Agency's list of medical cannabis products under the Danish Pilot Programme for medical cannabis, with import and distribution of the products Bedrocan®, Bedica®, and Bediol® to all the country's pharmacies.ĭanCann Pharma A/S (SS: DANCAN) (OTCQB: DCPXF) was founded in 2018 and is a Danish biopharmaceutical Company powered by cannabinoids. We have connected the desired commercial aspect to our Company, and it has already yielded results with the recently concluded distribution agreements with German WEECO Pharma and Polish Storkpharm, which together have a value of more than DKK 80 million distributed over the next three years," says the CEO of CannGros.ĬannGros ApS is approved for the production of cannabis products for medical use, in accordance with the law on the Danish Pilot Programme for medical cannabis. "CannGros has proven to be a really strong acquisition and an important addition to DanCann Pharma. Both in connection with the sale of own production, but also to a large extent through an upgrading and expansion of the activities in CannGros. For the time being, there will only be commercial and regulatory activities here," says Louise Conradsen, who also points out that being able to offer a workplace centrally in Denmark may make it easier to attract new colleagues, if that becomes relevant later on.Īt the end of 2022, DanCann Pharma received its EU-GMP certification from the Danish Medicines Agency and is therefore looking into a future where the commercial aspect will be more in focus. 
Recently, we moved the repackaging and distribution activities in CannGros from Glostrup to Ansager in order to have the group's activities consolidated at the headquarters in Southwest Jutland, and the opening of a new location in Odense does not change that. 
"CannGros was the first company to be approved by the Danish Medicines Agency for the import and handling of medical cannabis under the Danish Pilot Programme back in 2018. When the company acquired the wholesale company CannGros in the fall of 2021, DanCann Pharma became the market leader with license and sales rights for three available products on the market under the Danish Pilot Programme. Since its founding in 2018, DanCann Pharma has taken huge steps towards establishing itself as a producer and distributor of products on the market for medical cannabis. We have now found them in the old post office," says Louise Conradsen. "A large part of the Danish cannabis industry is located on Fyn, and we have for a long time been looking for suitable facilities in the local area. 
She is the CEO of the subsidiary CannGros ApS (“CannGros”) and sales manager of DanCann Pharma - and lives almost next door to the new office, located at Lille GråbrødrestrOdense C. Louise Conradsen will become the daily manager of the office in Odense. The office facilities in the old post office in the center of Odense will in the future form the framework for the Company’s commercial activities and work in the regulatory area. COPENHAGEN, Denmark, 23 February 2023 - DanCann Pharma A/S (SS: DANCAN) (OTCQB: DCPXF) ("DanCann Pharma" or the "Company"), a Danish company powered by cannabinoids, hereby announces that the Company on February 1st, opened a sales office centrally in Denmark. 
0 Comments
Read More
Leave a Reply. |
 RSS Feed
RSS Feed
